
BLOG ARTICLE
PK/PD Modeling and Simulation
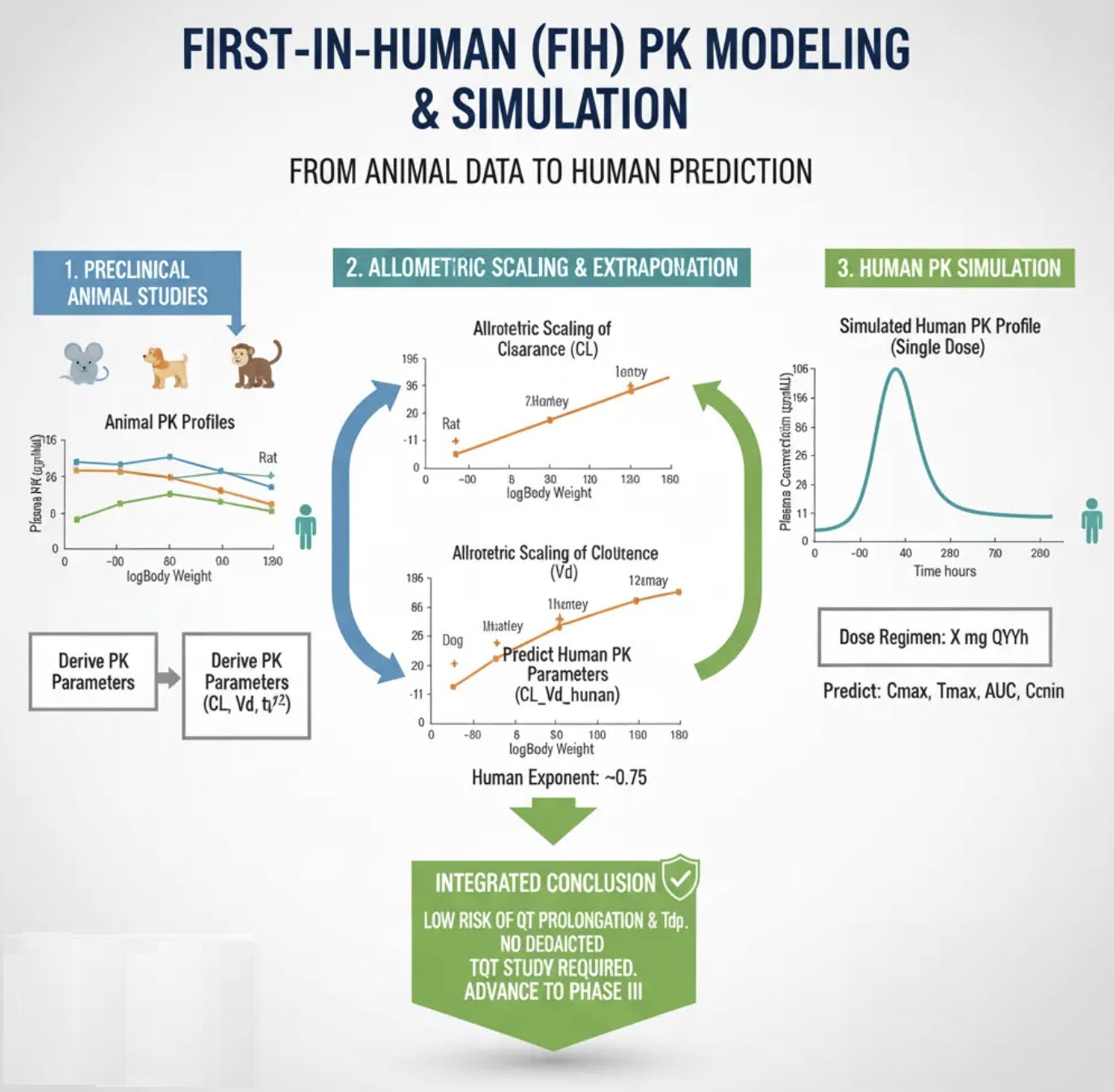
During FIH trial conduct, we performed non-compartment (NCA) PK analyses for each dose cohort on an ongoing basis. We found that the predicted and observed PK concentrations were in good agreement. The proposed dose range is adequate for RP2D selection.
©Copyright 2019 to 2026. All Rights Reserved