
BLOG ARTICLE
Thorough QT (TQT) Study Waiver: When Is It Justified and How to Achieve It?
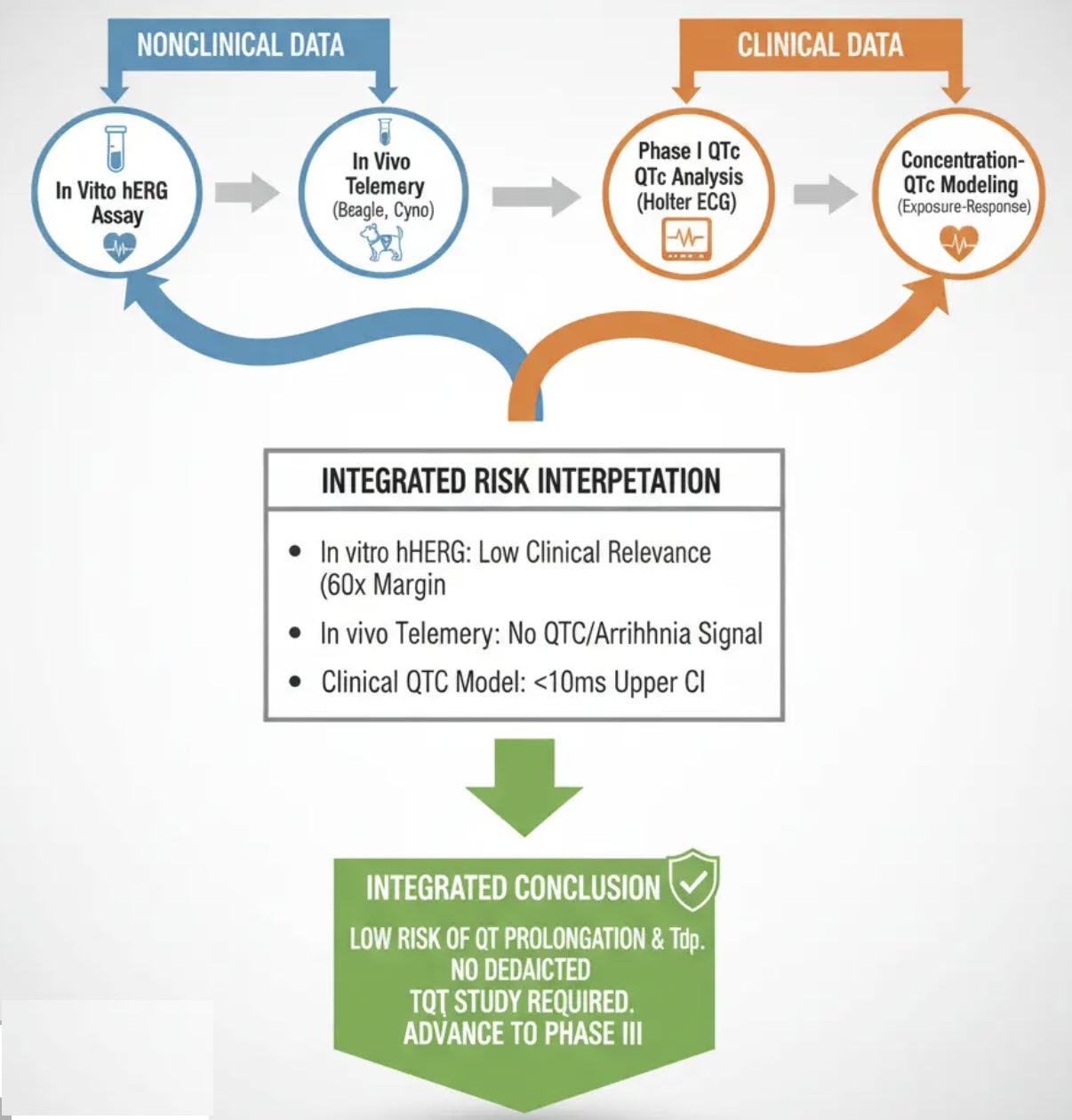
Regulatory agency agreed with the results of the integrated assessments conducted by XP Pharma. Client obtained the waiver for conducting a standalone TQT study.
©Copyright 2019 to 2026. All Rights Reserved